MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:

MLZ (eng)
Lichtenbergstr.1
85748 Garching
05.02.2021
Cell with windows: New design in lithium battery research

The scientists used a laser to cut the tiny windows, each with a diameter of 500 µm, into the cover of the coin battery. The small diameter of the laser-cut holes is necessary so that the contact pressure on the electrode stack remains sufficiently high for good electrochemical performance even in the area of the exit windows. © A. Heddergott / TUM
Scientists at the Technical University of Munich (TUM) have developed a new cell design for lithium-ion batteries for material characterization. The “battery with windows” makes it possible, using neutron scattering, to measure the lithium concentration within an electrode as a function of depth during charging and discharging. The new technique can help to elucidate both the reaction mechanisms and aging phenomena in batteries.
We encounter lithium-ion batteries everywhere in our everyday lives, whether in wristwatches, smartphones, laptops, or electric vehicles. Chemists and physicists at the Technical University of Munich (TUM) have now taken a closer look at the diagnostics for reaction
mechanisms in these energy carriers as part of an interdisciplinary research project.
To directly observe the operando distribution of the lithium concentration in battery electrodes, i.e., during charging and discharging, the scientists used the prompt gamma activation analysis (PGAA) facility at the research neutron source Heinz Maier-Leibnitz (FRM II) at TUM. Instrument scientist Dr. Zsolt Revay and PhD student Markus Trunk conduct experiments with Neutron Depth Profiling (NDP) there. “With NDP, samples can be analysed non-destructively, so the method is very suitable to our investigations,” explains Fabian Linsenmann, PhD student at the Chair of Technical Electrochemistry and first author of the study.
Neutron Depth Profiling in battery research

Fabian Linsenmann (right) prepares the air-sensitive lithium-ion cells in a glovebox for the experiment. Master's student Philip Rapp (left) tests the samples on a battery cycler before the NDP examination. © F. Linsenmann / TUM
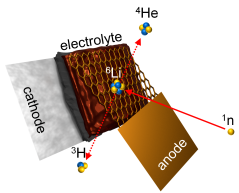
The principle of Neutron Depth Profiling at PGAA is based on the determination of the energy loss of charged particles in matter. Natural lithium consists of about 93% of the isotope 7Li and about 7% of 6Li. If 6Li is irradiated with cold neutrons, these can be captured by the lithium nuclei. The nuclei then decay and two charged particles with defined energies are produced, tritium (3H+) and alpha-particles (4He2+).
As they pass through matter, the particles lose energy. A detector can then subsequently measure the remaining energy of the particles, and thus scientists can trace the exact point of origin of the particle in the battery cell. This yields information about the amount and distribution of lithium in the battery. “The challenge with this method is that the charged particles lose their energy very quickly,” Linsenmann elaborates. Commercial battery cells would not be eligible for an NDP study, “because the walls of any normal battery cell are so thick that the particles would not reach the detector.”
New design – new insights
The scientists from the Physics Department and Chair of Technical Electrochemistry have therefore come up with something special: A coin cell battery, as often found for example in a wristwatch, with small windows that are permeable for charged particles, but which still behaves electrochemically like a conventional lithium-ion cell. “We were the first to actually show that the new cell design we developed for our NDP investigation is comparable to a normal battery in terms of electrochemical performance,” says Linsenmann, explaining a key component of the research project.
New cell design provides insight into battery fatigue
The scientists were thus able to gain insightful knowledge about the properties of the so-called “Solid Electrolyte Interface” (SEI). This layer forms in lithium-ion batteries during initial charging at the interface between the graphite particles, which make up the cell’s anode, and the electrolyte. It protects the electrolyte from being decomposed during further charging and discharging of the cell. The formation of the SEI binds a part of the available lithium in the battery and thus entails an irreversible loss of capacity.
When quantitatively comparing the amount of lithium in the SEI observed by NDP and the one calculated by charge/discharge capacity, things got exciting: The lithium concentration observed during the experiment was significantly lower than expected. A possible explanation for this would be that a significant part of the lithium-containing compounds in the SEI is soluble in the electrolyte, and thus diffuses into the electrolyte-filled interface between the anode and the cathode during the duration of the experiment (about 24 hours).
The basic findings and the newly developed cell design will be useful in future studies by helping to better elucidate, measure, and understand aging mechanisms in batteries.
Original publication:
Fabian Linsenmann, Markus Trunk, Philip Rapp, Lukas Werner, Roman Gernhäuser, Ralph Gilles, Bastian Märkisch, Zsolt Révay and Hubert A. Gasteiger. A Liquid Electrolyte-Based Lithium-Ion Battery Cell Design for Operando Neutron Depth Profiling (2020). Journal of the Electrochemical Society, Volume 167, Number 10. DOI: 10.1149/1945-7111/ab9b20
More information:
The research project was funded by the German Federal Ministry of Education and Research (BMBF), as part of the “ExZellTUM II” project (funding code 03XP0081) and the “N4DP” project (funding code 05K16WO1).
Related News
MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media: