MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:

MLZ (eng)
Lichtenbergstr.1
85748 Garching
11.10.2019
Perfectly suited: New ways in battery research
In the last years, lithium ion batteries have spread widely, in particular in mobile applications and in the electromobility sector. New concepts and materials emerge rapidly. These need to be characterized and their interplay needs to be understood in order to increase the efficiency and the lifespan of batteries built upon them. A collaboration of the Heinz Maier-Leibnitz Zentrum together with the Technical University of Munich is implementing a new method at the FRM II in Garching which is perfectly tailored to research on lithium-ion batteries: Neutron Depth Profiling.
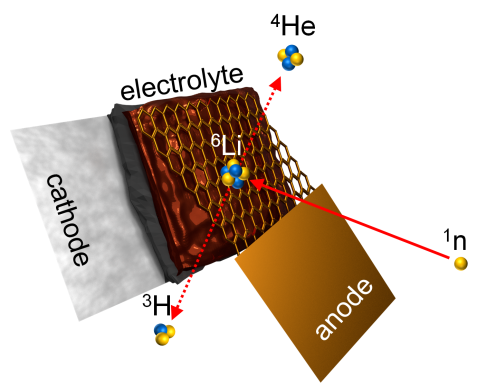
Principle of the neutron depth profiling: Lithium captures a neutron and decays into tritium and helium-4. With measuring the energy of the particle emerging from the surface, scientists can determine the original location of the lithium atom. © Markus Trunk, FRM II / TUM
New method for investigating lithium-ion batteries
The new instrument named “N4DP” is set up at the PGAA facility and makes use of the extremely high neutron capture cross sections of specific nuclei. Low-energy neutrons are captured by those nuclei in the sample which subsequently decay into two charged particles. The particle type and their respective energies are kinematically well defined and depend on the capturing nucleus. Since charged particles lose energy when traveling through matter, one can determine the depth of their origin by measuring their residual energy when leaving the sample. This allows for the precise determination of their density distribution in the sample. In contrast to other multi-element techniques of activation analysis (PGAA, NAA), neutron depth profiling is suitable for a couple of isotopes. Lithium-6 is one of the most prominent ones, which is the reason why NDP is particularly well suited for lithium-ion-battery research. The figure below shows the principle of NDP in the case of lithium-6. Using this method ex situ measurements are possible, where electrodes are removed from batteries after charging to different states-of-charge or after different numbers of charge-discharge cycles and are then exposed to the high neutron flux at the N4DP focal point. Additionally, the method also allows for operando measurements where the whole battery can be analyzed during the battery charging and discharging process.
Lithium-ion batteries using silicon-graphite electrodes
They characterized a promising new electrode candidate for silicon-graphite electrodes which has a silicon fraction of 35%. However, this silicon-based anode electrode also suffers from a loss in capacity with the number of charge-discharge cycles. The scientists in this cooperation identified two reasons for this: firstly, upon extended charge-discharge cycling the anode electrode substantially expands in its thickness and increases in mass. The material for this mass gain is provided by the electrolyte which undergoes side reactions at the anode electrode with the electrolyte, forming a so-called SEI layer (“solid electrolyte interphase”). Secondly, morphological changes of the silicon nanoparticles largely increase their surface area, which further increases the SEI formation. Both results were confirmed by the research group using conventional electrochemical and imaging techniques. Using NDP, they could go one step further for the first time and take a detailed look at the electrode mass increase. They realized that the increasing amount of lithium immobilized in the SEI with increasing numbers of charge-discharge cycles proceeds uniformly across the thickness of the silicon-graphite electrode. This is an important finding, because the scientists can now exclude ionic and/or electrical transport limitations across the electrode thickness as the possible cause for the observed capacity loss upon extended cycling. Thus, the observed degradation is not due to clogging of the electrode pores by SEI products, but is predominantly caused by the volume expansion/contraction of silicon nanoparticles over a charge-discharge cycles as well as by the gradual surface area increase caused by the morphological changes of the silicon nanoparticles.
Depth-resolved measurements during charging and discharging
Furthermore, it is possible to go beyond the previous ex situ measurements of the SEI. “The possibility of a depth-resolved operando measurement enables us to go further than just a characterization of the passive SEI layer. We can also look into the reversible lithiation, which happens during the charging and discharging process.”, says Markus Trunk, part of the TUM collaboration, while preparing with his colleagues Morten Wetjen and Lukas Werner the samples for the next N4DP measurements.
Original publications:
Just lately the TUM cooperation – consisting of the MLZ, the professorship of particle physics at low energies, the chair of technical electrochemistry, and the central technology laboratory of the physics department – contributed significantly to the understanding of a new kind of lithium-ion batteries using the N4DP instrument. They published their recent results in three different papers:
M. Trunk, L. Werner, R. Gernhäuser, B. Märkisch, Z. Revay, H. Gasteiger, M. Wetjen, R. Gilles, Materials Characterization 146, 127 (2018), DOI: https://doi.org/10.1016/j.matchar.2018.09.030
M. Wetjen, M. Trunk, L. Werner, R. Gernhäuser, B. Märkisch, Z. Revay, R. Gilles, H. A. Gasteiger, Journal of the Electrochemical Society 165 (10), A2340 (2018), DOI: 10.1149/2.1341810jes
M. Wetjen, M. Trunk, L. Werner, H.A. Gasteiger, R. Gernhäuser, R. Gilles, B. Märkisch, Z. Revay, Journal of the Electrochemical Society 166(8), A1408 (2019), DOI: 10.1149/2.0581908jes
More information:
Neutron Depth Profiling
MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:


