MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:

MLZ (eng)
Lichtenbergstr.1
85748 Garching
02.08.2013
The engines of life can perform without water
Neutron experiments explain how protein-polymer hybrids work.
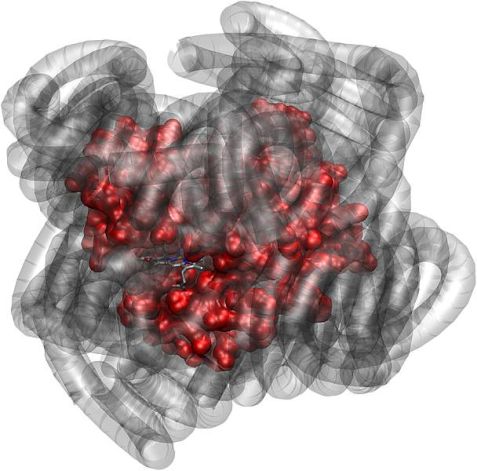
The protein molecule myoglobin (red) is biologically active, even if its natural water shell is replaced by a polymer shell (gray). (Source: J. Am. Chem. Soc., DOI 10.1021/ja303894g)
Proteins do not need to be surrounded by water to carry out their vital biological functions, according to an international team of scientists. The researchers used state of the art neutron scattering techniques to demonstrate that when myoglobin, an oxygen-binding protein found in the muscle tissue of vertebrates, is enclosed in a sheath of surfactant molecules, it moves in the same way as when it is surrounded by water. These motions are essential if a protein is to perform its biological function, and their findings make proteins a viable material for use in new wound dressings or even as chemical gas sensors.
Water is the natural environment for soluble proteins and an integral part of their structures which allows them to carry out their specific function. It had been perceived for many years that proteins required water or another solvent in order to function. But in 2010 researchers proved that by grafting polymer chains onto the protein surface, it was possible to produce solvent- and water-free myoglobin liquids that could still perform their biological roles. Scientists have now demonstrated that protein dynamics is the reason why.
Myoglobin is common to almost all mammals and responsible for the red colour of raw meat. Like all soluble proteins, its surface is covered with water molecules. In this study a team of researchers from France, UK, Australia and Germany wanted to assess whether the protein structure could still move and continue to bind oxygen if all the water was completely removed and replaced by synthetic molecules.
The team analysed three samples, a wet sample (the protein in water), a dry sample (the dehydrated protein) and a dry protein-polymer hybrid sample where the water molecules had been replaced by synthetically crafted polyethylene glycol-based polymer surfactant molecules. Using a technique called incoherent neutron scattering at instruments of Forschungszentrum Jülich at the Research Neutron Source Heinz Maier-Leibnitz (FRM II) in Garching near Munich, Germany, and at instruments of the Institut Laue-Langevin (ILL) in Grenoble, France, the team was able to monitor the motions in the protein and in the polymer surfactant separately. What they found was that the myoglobin molecules surrounded by polymer moved just as well as the wet sample, and that the dry sample had very little mobility. “It works like this only with Neutrons”, says Dr. Joachim Wuttke from Jülich Centre for Neutron Science (JCNS) in Garching, who helped the research team with their experiments at the instrument.
press release of the Institut Laue-Langevin (ILL)
Original Publications:
A polymer surfactant corona dynamically replaces water in solvent-free protein liquids and ensures macromolecular flexibility and activity;
Gallat et al. ;
J. Am. Chem. Soc.
DOI, 10.1021/ja303894g
Dynamical Coupling of Intrinsically Disordered Proteins and Their Hydration Water: Comparison with Folded Soluble and Membrane Proteins;
Gallat et al.;
Biophysical Journal, Volume 103, Issue 1, 3 July 2012,
Pages 129–136;
DOI: 10.1016/j.bpj.2012.05.027
MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:


