MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:

MLZ (eng)
Lichtenbergstr.1
85748 Garching
28.03.2018
Ping-pong in the crystalline tunnel

Prof. Dr. Alois Kuhn (left) and Dr. Markus Hölzel at the structure powder diffractometer SPODI at MLZ. © Wenzel Schürmann / TUM
Lithium-ion batteries are an indispensable part of our daily lives. The negative electrodes are typically based on graphite. However, they have some problems, such as the so-called lithium plating. Now there is an electrode material that is not based on graphite and does not show these disadvantages: sodium hexatitanate.
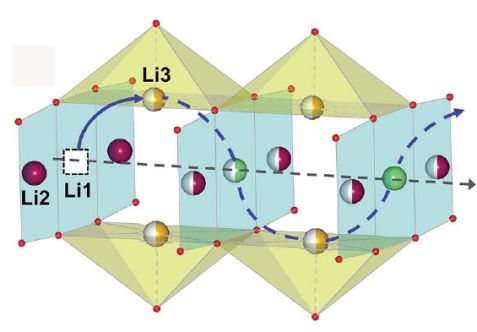
Sodium hexatitanate has high structural stability and is therefore a promising material for long life of batteries. Batteries with these negative electrodes, the anodes, can be charged quickly with high voltage and, in this case, no lithium layer is formed on the anode material in this case, therefore the risk of short circuits and explosions is much lower. Sodium hexatitanate has a very special open structure that opens a tunnel to the lithium ions. This results in the potential of lithium storage as well as the mobility of lithium ions during charging and discharging.
But how exactly does the mechanism of lithium storage work here? There were ideas, but there was no experimental proof.
A Spanish-German research team has now taken a close look at the properties of this anode material in order to answer the question of how lithium behaves in this structure. For this they tried electrochemical methods, synchrotron experiments in the ESRF in Grenoble, solid-state NMR spectroscopy at the Institute of Materials Science of Madrid and neutron diffraction studies on the MLZ instrument SPODI with Dr. Markus Hölzel. “A decisive factor in this extensive investigation was the neutron diffraction, which allowed us to determine the exact positions of lithium”, says project leader Prof. Dr. Alois Kuhn from the University of CEU San Pablo in Madrid.
How does the mechanism work? Part of the lithium ions occupy the inside of the tunnel, which is formed by the structure of sodium hexatitanate. The structure distinguishes different positions for the lithium ions, some are on the sidewalls, others on the tops or bottoms of the tunnel, others in the middle. Some of these lithium ions are mobile and in their motion repelled by anchored sodium and lithium ions. This repulsion ensures that the free lithium ions are propelled like ping-pong balls between the anchored ions.
“These were really extensive studies and the combination of all these methods helped us to make a breakthrough,” says a delighted Alois Kuhn. “Of course, the approach chosen here can also be applied to other battery materials and thus creates the opportunity to optimize such materials.” As a German professor at a Spanish university, Kuhn is extremely grateful that he and other scientists receive access to Heinz Maier -Leibnitz center. He makes extensive use of this, because he will soon come to Garching for further experiments.
Original publication:
Alois Kuhn, Juan Carlos Pérez-Flores, Markus Hoelzel, Carsten Baehtz, Isabel Sobrados, Jesús Sanz and Flaviano García-Alvarado
Comprehensive investigation of the lithium insertion mechanism of the Na2Ti6O13 anode material for Li-ion batteries
J. Mater. Chem. A 6, 443
MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media: