MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media:

MLZ (eng)
Lichtenbergstr.1
85748 Garching
02.07.2020
Neutrons make reaction pathways of important enzymes visible for the first time

Dr. Andreas Ostermann adjusts a crystal for neutron measurement at the BioDiff. © Andreas Heddergott
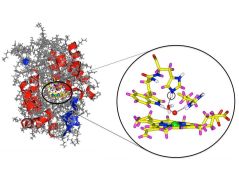
An international team of researchers has now used neutrons to visualize the proton transfer in the enzyme ascorbate peroxidase for the first time in detail and suggested reaction pathways.
Citrus fruits are particularly rich in ascorbic acid, often also called vitamin C. The vitamin not only performs important services in the human body. In plant cells, the enzyme ascorbate peroxidase also needs vitamin C to convert the cell toxin hydrogen peroxide into water.
In this reaction, protons and electrons must be transferred from ascorbic acid to hydrogen peroxide with the help of the enzyme. Ascorbate peroxidase is an example of a whole range of enzymes that also play a decisive role in many vital reactions in the human body through the transfer of protons and electrons.
Neutrons show the hydrogen atom positions
“Due to a lack of suitable measuring methods, it was previously unclear exactly which path the protons take in the enzyme and in what state the ascorbate is present during the reaction,” says the head of the study, Prof. Emma Raven from Bristol University.
Until now, enzymes have mainly been analysed using X-rays. However, very light atoms, such as hydrogen, are almost invisible to X-rays. Neutrons, on the other hand, are also sensitive to light atoms. Therefore, the British team had the samples analysed at the Heinz Maier-Leibniz Zentrum (MLZ) at the Technical University of Munich (TUM) and the Institut Laue-Langevin (ILL) in Grenoble (France).
Surprising: neutral instead of positive
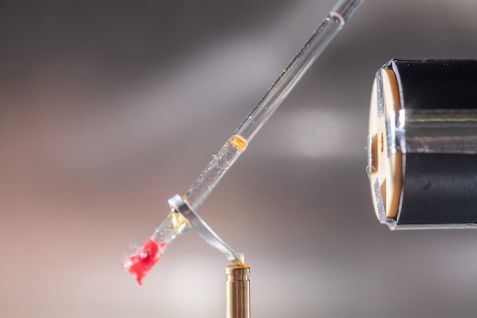
The BioDiff instrument enables scientists to study the structure of biological molecules with the help of neutrons. © Andreas Heddergott
The team brought a crystal of the enzyme ascorbate peroxidase with bound vitamin C to the MLZ in Garching, where scientists from the Heinz Maier-Leibnitz Zentrum (MLZ) analysed it. As a reference structure, they had an enzyme crystal without bound vitamin C examined at the Institute Laue-Langevin.
Dr. Tobias Schrader (Forschungszentrum Jülich) and Dr. Andreas Ostermann (Technical University of Munich) carried out the measurement on their instrument BIODIFF. The structural model obtained from the measurement data surprised the British biochemists.
“A special part of the enzyme, an arginine amino acid building block, was present in a neutral form,” says Prof. Peter Moody from the University of Leicester, who co-led the work. “Until now it had been assumed to be positively charged.” Therefore, the enzyme is missing a proton at this point. This in turn suggests that a proton might have migrated from vitamin C to the active centre via this pathway.
Findings also transferable to other enzyme reactions
Nearby, there is another candidate in the active centre, a histidine amino acid building block, which can take up or release a proton. Both together could form the basis for a general mechanism for proton transport.
The scientific team envisages a third way to transport protons to the active centre of the enzyme, which is a chain of water molecules. Although these are relatively far apart, they could also transport protons by changing their position slightly.
“Using neutron scattering, we have identified three possible routes by which a proton required for the reaction can reach the active centre of the enzyme,” says Emma Raven. “Ascorbate peroxidase belongs to the large group of haemenzymes. These results also help us to better understand similar reactions in other enzymes of this family.”
Original publication:
Visualizing the protons in a metalloenzyme electron proton transfer pathway. Hanna Kwon, Jaswir Basran, Juliette M. Devos, Reynier Suardiaz, Marc W. van der Kamp, Adrian J. Mulholland, Tobias E. Schrader, Andreas Ostermann, Matthew P. Blakeley, Peter C. E. Moody, Emma L. Raven. PNAS, first published March 9, 2020. DOI: 10.1073/pnas.1918936117
More information:
The research was funded by the Biotechnology and Biological Sciences Research Council and the Engineering and Physical Sciences Research Council under the UK Catalysis Hub. Neutron radiation for the determination of crystal structures was provided by the Research Neutron Source Heinz Maier-Leibniz (FRM II) in Garching and the Institut Laue-Langevin in Grenoble (France). Computing time was provided by the Advanced Computing Research Centre of the University of Bristol and the ARCHER UK National Supercomputing Service.
Related News
-
10.07.2014
Freeze-frame of an enzyme
MLZ is a cooperation between:
 > Technische Universität München
> Technische Universität München > Helmholtz-Zentrum Hereon
> Helmholtz-Zentrum Hereon
 > Forschungszentrum Jülich
> Forschungszentrum Jülich
MLZ is a member of:
 > LENS
> LENS > ERF-AISBL
> ERF-AISBL
MLZ on social media: